The newly published study describes a breakthrough that feels almost like watching human bone marrow come alive in a dish. Researchers built a realistic bone marrow model using human stem cells, and it behaves like the real tissue that keeps our blood system running. This matters now because blood cancers, immune disorders, and transplant failures often begin in this fragile microenvironment, and scientists have struggled to study it directly in humans.
Study: New human bone marrow model built with stem cells. Focus: How blood stem cells grow and behave. Why It Matters: Helps scientists study blood cancers, immune disorders, and future therapies in realistic human settings.
The team created what they call an engineered vascularized osteoblastic niche, a fully human bone marrow–like system that mimics the endosteal niche. This niche is the region inside bone where blood stem cells rest, repair, and wake up when the body needs new immune or blood cells. Earlier organoids could not recreate this space because they were too small, lacked real bone-like material, or depended on animal-derived ingredients. This new model breaks those limits by using human induced pluripotent stem cells, a porous hydroxyapatite scaffold that resembles real bone, and a guided development process that allows the tissue to organize itself naturally.
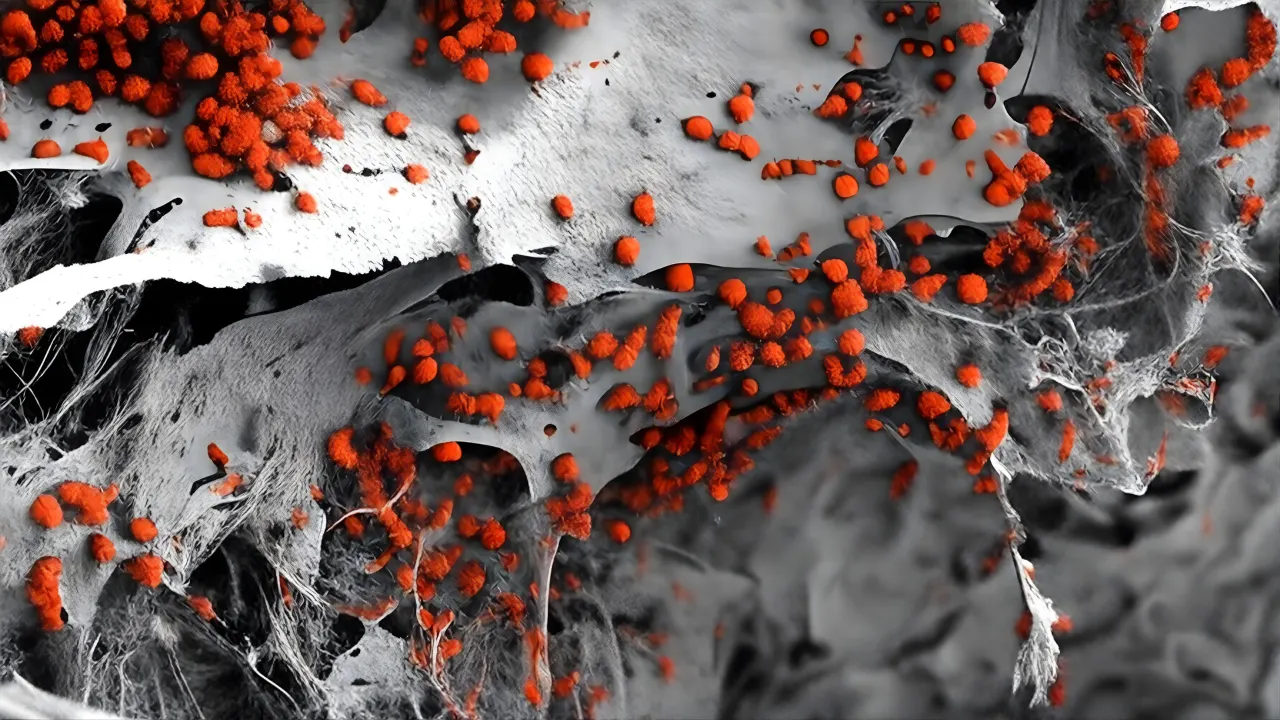
To prove the model worked, the researchers grew two types of organoids: one forming blood vessels and another forming bone-producing cells. They joined these inside the scaffold and watched them fuse into a single living tissue. High-resolution images showed tiny blood vessels forming naturally, covered with supportive pericytes and surrounded by bone-like matrix. They used simple imaging, molecular tests, and electron microscopy to confirm that the vessels looked and behaved like those in human marrow. This included fenestrations, tip cells, and capillaries measuring five to thirty microns, similar to those seen in the endosteal region.
The discovery matters because the niche controls how blood stem cells behave. When the researchers added human blood stem and progenitor cells, the engineered marrow kept them alive, allowed them to mature, and even preserved their ability to repopulate blood systems when transplanted into mice. The vascularized version of the model boosted early myeloid development, suggesting that real blood vessels play a major role in steering stem cell decisions. Genes linked to healthy stem cell support, like CXCL12 and VCAM1, were more active, explaining why the system performed better than avascular controls.
Experts say this model gives the field its first chance to watch human marrow behave in controlled conditions. The research team noted that genes involved in aggressive leukemias, including those linked to chemoresistance, were also increased. That means the model may help show why some cancers hide in bone marrow niches and resist treatment. Some specialists caution that organoids cannot yet replace full bone physiology, but agree the system is a major step forward because it stays stable for weeks, develops neural and immune-like cells on its own, and reacts predictably to molecular signals.
This breakthrough connects to larger issues in medicine and biotechnology. Better marrow models could help improve stem cell transplants, test drugs for leukemia, study aging-related immune decline, and reduce reliance on animal research. The results also show how human tissue engineering is merging with regenerative medicine, cancer biology, and personalized therapy. These engineered niches could eventually be matched to individual patients to predict how their cells will behave.
Next, the researchers plan to refine the model by controlling niche factors such as CXCL12 or VEGF and by testing how cancer cells reshape the tissue. They also hope to use genetic tools to switch signals on and off inside the niche. Uncertainties remain about how long the system can remain stable, why certain cell line variations produce different vascular shapes, and how closely it will mimic late-stage diseases.
The study offers a clear takeaway: scientists have finally built a human bone marrow model that captures how real niches support and guide blood stem cells. This opens a direct, human-focused window into one of the most complex and hidden tissues in the body.
Story Source:
Materials provided by University Hospital Basel and collaborating institutions. Content may be edited for style and length.
Journal Reference:
Qing Li, Marina T. Nikolova, Gangyu Zhang, Igor Cervenka, Federica Valigi, Dominik Burri, Evelia Plantier, Andrea Mazzoleni, Anais Lamouline, Juerg Schwaller, Barbara Treutlein, Ivan Martin, Andres García-García. Macro-scale, scaffold-assisted model of the human bone marrow endosteal niche using hiPSC-vascularized osteoblastic organoids. Cell Stem Cell, 2025. Volume 32(December Issue). DOI: https://doi.org/10.1016/j.stem.2025.10.009
Focus Keyword:
human bone marrow model