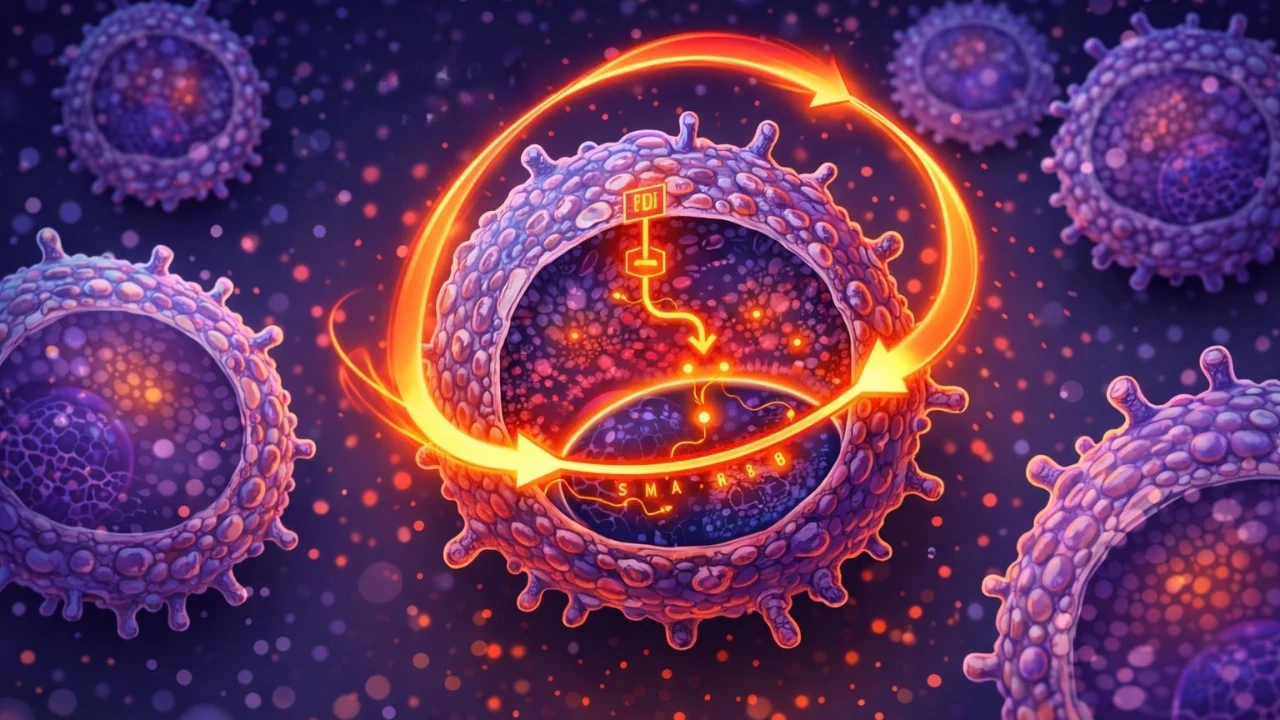
Researchers in In Hwa Jang’s lab at the University of Minnesota Medical School discovered an unexpected feedback loop inside aging immune cells. As macrophages grow older, they begin producing a protein called GDF3. Instead of shutting down after doing its job, GDF3 sends signals back to the very cells that released it, keeping them stuck in an inflammatory state through a pathway known as SMAD2/3.
This self-reinforcing cycle offers a new explanation for inflammaging, the chronic inflammation that rises with age. The study, published in Nature Aging, was led by Christina Camell, PhD, and linked to real-world health data from the ARIC study through collaboration with Pamela Lutsey, connecting lab discoveries to population health.
Inside this loop, GDF3 essentially rewires the macrophages from within. It alters how their DNA is packaged and read, locking the cells into long-lasting inflammatory behavior. The finding has drawn attention beyond the lab, appearing in Nature Aging and being highlighted by outlets like ScienceDaily and the University of Minnesota Medical School, signaling its growing importance in aging and immune research.
From Lab Bench to the Clinical Frontier
The story moves from mechanism to meaning: aging macrophages keep inflammatory signals alive through the GDF3-SMAD2/3 axis, a discovery that translates mechanistic biology into a testable therapeutic target. In preclinical aging models, blocking this signaling reduced harmful inflammation and yielded meaningful improvements in survival, supporting a direct route to human benefit once clinical trials prove safety and efficacy.
What Happens If We Block GDF3-SMAD2/3?
The normal, mature immune response risks tipping into chronic, damaging inflammation as we age. By interrupting the feedback loop, researchers can lower inflammatory markers and potentially blunt the risk of deadly inflammations such as sepsis in older adults. The path forward hinges on human studies to verify safety, dosing, and long-term effects—but the signal is clear: this axis is a tangible therapeutic target.
Looking Ahead: A New Era for Aging Health
Today’s work, bolstered by ARIC data and collaboration with Pamela Lutsey, opens a translational corridor from molecule to medicine. If clinical trials confirm safety and efficacy, the GDF3-SMAD2/3 axis could become a central lever for reducing inflammaging and improving metabolic health in older adults, shifting sepsis risk and chronic disease trajectories. The era of chronic immune alarm in aging could be re-written as therapies move toward real-world use.
- GDF3 drives a self-sustaining inflammatory loop in aging macrophages via SMAD2/3.
- Blocking GDF3-SMAD2/3 in preclinical models reduced inflammation and improved survival, signaling therapeutic potential.
- Translation to humans requires trials, but the pathway offers a concrete target for aging-related immune health.
Why it matters: inflammaging is a top public-health concern. Targeting the GDF3-SMAD2/3 axis could reduce sepsis risk, improve metabolic health, and extend healthy lifespan—and it arrives at a moment when the public conversation about aging and immunity is accelerating. For researchers, clinicians, and patients, this is a rare moment when a dry mechanism becomes a practical medical frontier.
Just 150 minutes of weekly exercise may help keep your brain biologically younger, according to new research on brain aging.
Read the Full Story